
RECENT PUBLICATIONS
Mechanistic insight into the microtubule and actin cytoskeleton coupling through dynein-dependent RhoGEF inhibition.
Meiri D, Marshall CB, Greeve MA, Kim B, Balan M, Suarez F, Bakal C, Wu C, Larose J, Fine N, Ikura M, Rottapel R.
Mol Cell. 2012 Mar 9;45(5):642-55.
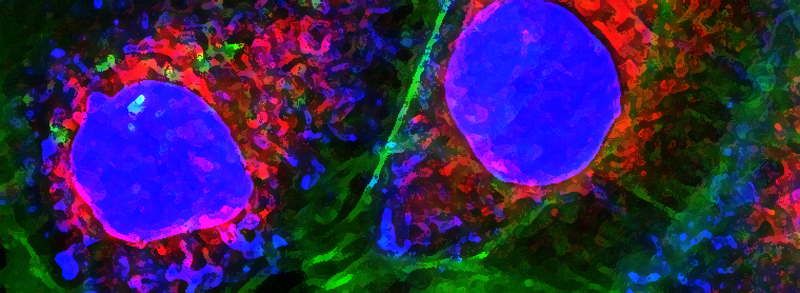
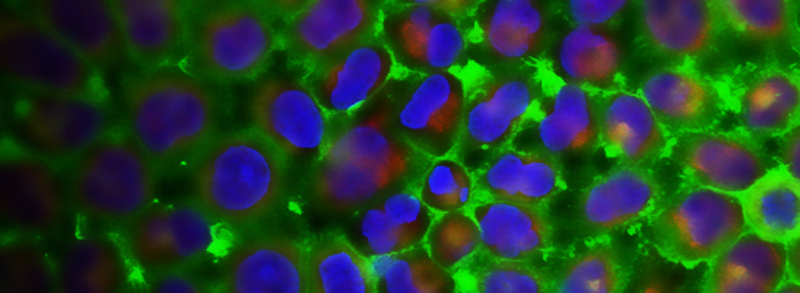
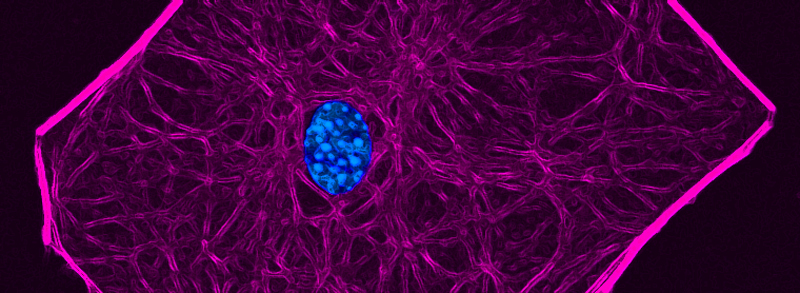
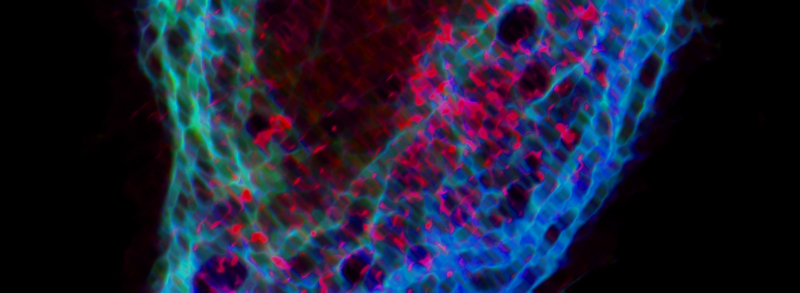
Actin-based stress fiber formation is coupled to microtubule depolymerization through the local activation of RhoA. While the RhoGEF Lfc has been implicated in this cytoskeleton coupling process, it has remained elusive how Lfc is recruited to microtubules and how microtubule recruitment moderates Lfc activity. Here, we demonstrate that the dynein light chain protein Tctex-1 is required for localization of Lfc to microtubules. Lfc residues 139-161 interact with Tctex-1 at a site distinct from the cleft that binds dynein intermediate chain. An NMR-based GEF assay revealed that interaction with Tctex-1 represses Lfc nucleotide exchange activity in an indirect manner that requires both polymerized microtubules and phosphorylation of S885 by PKA. We show that inhibition of Lfc by Tctex-1 is dynein dependent. These studies demonstrate a pivotal role of Tctex-1 as a negative regulator of actin filament organization through its control of Lfc in the crosstalk between microtubule and actin cytoskeletons.
